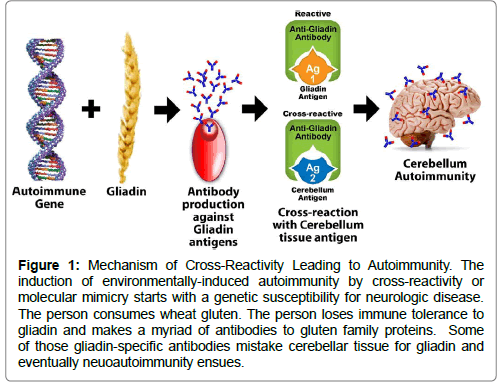
Valency of AMPA-IgM was crucial for antigen recognition as PTM-reactivity significantly decreased when AMPA-IgM were expressed as IgG. PTM antigen recognition was still present, although reduced, after reverting the IgM into germline. Pentameric monoclonal and polyclonal AMPA-IgM displayed cross-reactivity to multiple antigens and different PTMs. 
The antibodies were analysed on a panel of PTM antigens and tested for complement activation. Subsequently, pentameric monoclonal AMPA-IgM, their germline counterparts and monomeric IgG variants were generated. AMPA-IgM B cell receptor sequences were obtained by single cell isolation using antigen-specific tetramers. Polyclonal AMPA-IgM were isolated from RA patients and assessed for cross-reactivity towards PTM antigens. Moreover, we investigated whether AMPA-IgM can efficiently recruit immune effector mechanisms. We now studied the reactivity of (germline) AMPA-IgM to further understand the breach of B cell tolerance and to identify if cross-reactivity depends on extensive SHM. Although AMPA-IgG cross-reactivity to multiple post-translational modifications (PTMs) is evident, it is unknown whether the first responding B cells, expressing IgM, display similar characteristics or if cross-reactivity is crucially dependent on somatic hypermutation (SHM). Anti-modified protein antibodies (AMPA) targeting citrullinated, acetylated and/or carbamylated self-antigens are hallmarks of rheumatoid arthritis (RA).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
